
CasNo: 13601-19-9
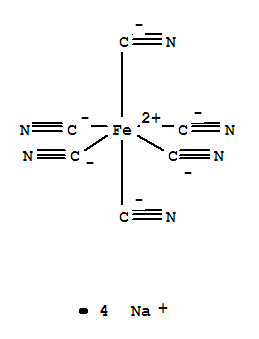
Molecular Formula: Na4Fe.(CN)6
Appearance: Odorless yellow solid
|
Production |
Sodium ferrocyanide is produced industrially from hydrogen cyanide, ferrous chloride, and calcium hydroxide, the combination of which affords Ca2[Fe(CN)6] ? 11H2O. A solution of this salt is then treated with sodium salts to precipitate the mixed calcium-sodium salt CaNa2[Fe(CN)6], which in turn is treated with sodium carbonate to give the tetrasodium salt. |
|
Storage |
Hygroscopic, -20°C Freezer, Under inert atmosphere |
|
Reactivity |
Sodium ferrocyanide is a coordination compound of iron. The cyanide ligands are tightly bound to the iron, so it is not as toxic as simple inorganic cyanide salts. However, this compound can release hydrogen cyanide gas upon reaction with acids. |
|
Air & Water Reactions |
Water soluble. |
|
Reactivity Profile |
Sodium ferrocyanide has weak oxidizing or reducing powers. Redox reactions can however still occur. The majority of compounds in this class are slightly soluble or insoluble in water. If soluble in water, then the solutions are usually neither strongly acidic nor strongly basic. These compounds are not water-reactive. |
|
Health Hazard |
None recorded. |
|
Flammability and Explosibility |
Nonflammable |
|
Safety Profile |
When heated to decomposition emits toxic fumes of CN-. |
|
Purification Methods |
Crystallise it from hot water (0.7mL/g), until free of ferricyanide as shown by the absence of formation of Prussian Blue colour with ferrous sulfate solution. |
|
Chemical properties |
Sodium ferrocyanide is the sodium salt of the coordination compound of formula [Fe(CN)6]4?. In its hydrous form, Na4Fe(CN)6 ? 10H2O (sodium ferrocyanide decahydrate), it is sometimes known as yellow prussiate of soda. It is a yellow crystalline solid that is soluble in water and insoluble in alcohol. The yellow color is the color of ferrocyanide anion. Despite the presence of the cyanide ligands, sodium ferrocyanide has low toxicity (acceptable daily intake 0–0.025 mg/kg body weight). The ferrocyanides are less toxic than many salts of cyanide, because they tend not to release free cyanide.However, like all ferrocyanide salt solutions, addition of an acid can result in the production of hydrogen cyanide gas, which is toxic. |
|
Applications |
Sodium hexacyanoferrate(II) decahydrate is being used and studied as a possible cathode material for sodium ion batteries. |
|
appearance |
Sodium ferrocyanide is a yellow crystalline salt Na4Fe(CN)6 similar to potassium ferrocyanide and used in making iron blue pigments, blueprint paper, and dyes — Called also yellow prussiate of soda |
|
General Description |
Odorless yellow solid. Sinks and mixes with water. |
InChI:InChI=1/12CN.2Fe.4Na/c12*1-2;;;;;;/q;;;;;;;;;;;;2*-2;4*+1/r2C6FeN6.4Na/c2*8-1-7(2-9,3-10,4-11,5-12)6-13;;;;/q2*-2;4*+1
CAS:30123-17-2
CAS:95-45-4
CAS:9004-32-4